|
12/28/2023 0 Comments S block
This is due to the presence of the loosely held valence electron which can easily be removed. The s-block elements are good conductors of heat and electricity. This is because of the increase in atomic size and increasing ease with which an electron can be removed from the outermost shell. The reactivity of s-block elements increases down the group. Therefore, they can form mono-positive and dipositive cations. These electrons can be easily removed due to the low value of ionization potential. The s-block elements contain one or two electrons in their outermost shell. The melting and boiling points of alkaline earth metal are shown below: 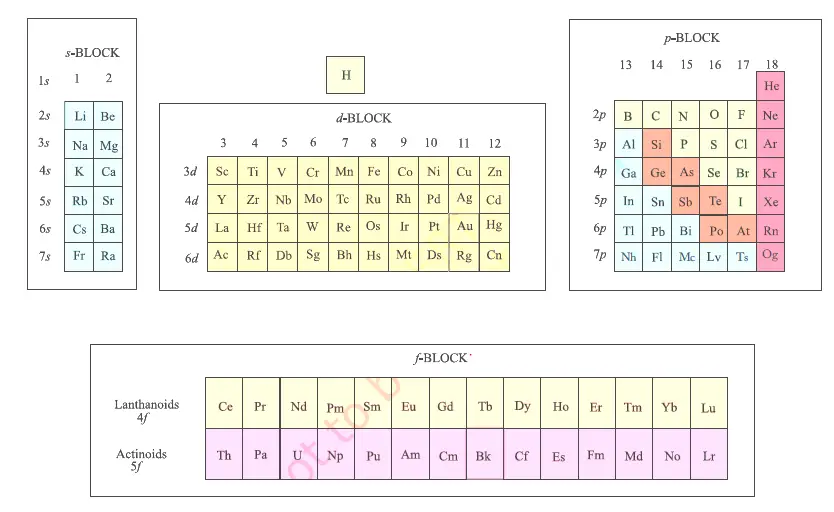
The melting and boiling points of alkali metals are shown below: Similarly, their melting and boiling points decrease down the group due to an increase in size. The s-block elements have low melting and boiling points due to their large sizes.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
